Opening Statement Chairman Dan Burton
Committee on Government Reform
“The Status of Research into Vaccine
Safety and Autism”
June 19, 2002
2154 Rayburn House Office Building
11:00 a.m.
In April the Committee conducted a hearing
reviewing the epidemic of autism and the Department of Health and Human Service’s
(HHS) response. Ten years ago, autism was
thought to affect 1 in 10,000 individuals in the United States. When the Committee began its oversight
investigation in 1999, autism was thought to affect 1 in 500 children. Today, the National Institutes of Health (NIH)
estimates that autism affects 1 in 250 children.
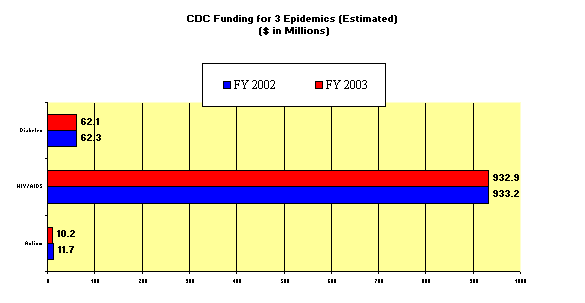
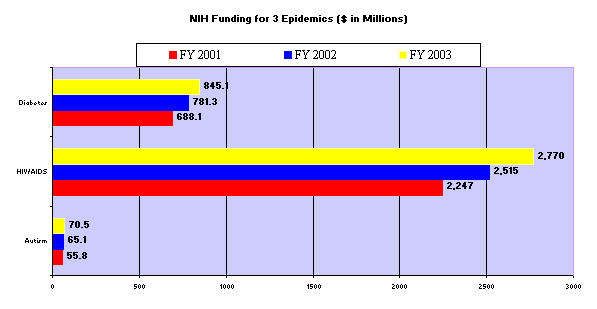
In April we
looked at the investment our Government has made into autism as compared to other
epidemics. We showed in that hearing that the
CDC and NIH have not provided adequate funding to address the issues in the manner that
our Public Health Service agencies have used to address other epidemics.

After our
hearing, I joined with my colleagues on the Coalition on Autism Research and Education to
request from our appropriators that at least 120 million dollars be made available in FY
2003 for autism research across the NIH and at that an additional $8 million be added to
the CDC’s budget for autism research.
Giving more
money to research is not the only answer though. Oversight
is needed to make sure that research that is funded will sufficiently answer the questions
regarding the epidemic, how to treat autism, and how to prevent the next ten years from
seeing the statistic of 1 in 250 from becoming 1 in 25 children.
High quality
clinical and laboratory research is needed now, not five or ten years from now. Independent analysis of previous epidemiological
and case control studies is needed as well.
We have learned that a
majority of parents whose children have late-onset or acquired autism believe it is
vaccine-related. They deserve answers. We have also learned that the parents have been
our best investigators in looking for both causes of autism and for treatments.
It has been parents who have
formed non-profit organizations to raise research dollars to conduct the research that the
CDC, the FDA, and the NIH have neglected to do. We
have heard from many of these parents in the past, Elizabeth Birt, Rick Rollens, Shelley
Reynolds, and Jeanna Smith, to name just a few. Each
of these parents had healthy babies who became autistic after vaccination.
I might have been like many
of the officials within the public health community – denying a connection - had I
not witnessed this tragedy in my own family. I
might not have believed the reports from parents like Scott and Laura Bono, Jeff Sell,
Jeff and Shelly Segal, and Ginger Brown, who came to me with pictures, videos and medical
records. I might have been like so many
pediatricians who discounted the correlation between vaccination and the onset of fever,
crying, and behavioral changes. Because both
of my grandchildren suffered adverse reactions to vaccines, I could not ignore the parent’s
plea for help. I could not ignore their
evidence.
My only grandson became
autistic right before my eyes – shortly after receiving his federally recommended and
state-mandated vaccines. Without a full
explanation of what was in the shots being given, my talkative, playful, outgoing healthy
grandson Christian was subjected to very high levels of mercury through his vaccines. He also received the MMR vaccine. Within a few days he was showing signs of autism.
As part of our
investigation, the Committee has reviewed ongoing concerns about vaccine safety, vaccine
adverse events tracking, the Vaccine Safety Datalink (VSD) Project, and the National
Vaccine Injury Compensation Program. I have
joined with Congressman Weldon, Congressman Waxman and 32 other members of Congress in
introducing HR 3741, the National Vaccine Injury Compensation Program Improvement Act of
2002 to realign the compensation program with Congressional Intent.
In today’s hearing, we
will receive a research update from several previous witnesses as well as new research
findings that further support a connection between autism and vaccine adverse events. We will learn more about both the possible link
between the use of the mercury-containing preservative thimerosal in vaccines and autism,
as well as autistic entercolitis resulting from the Measles-Mumps-Rubella (MMR) vaccine.
Through a Congressional
mandate to review thimerosal content in medicines, the FDA learned that childhood
vaccines, when given according to the CDC’s recommendations exposed over 8,000
children a day in the United States to levels of mercury that exceeded Federal guidelines. Is there a connection between this toxic exposure
to mercury and the autism epidemic? We will
hear from Dr. James Bradstreet and Dr. Vera Stejskal on this issue.
We have twice received
testimony from Dr. Andrew Wakefield regarding his clinical research into autistic
entercolitis. We will learn today that
not only has he continued to conduct clinical research, but that this research is
confirming the presence of vaccine-related measles RNA in the biopsies from autistic
children. Dr. Wakefield - like many
scientists who blaze new trails - has been attacked by his own profession. He has been forced out of his position at the
Royal Free Hospital in England. He and his
colleagues have fought an uphill battle to continue the research that has been a lone ray
of hope for parents whose children have autistic entercolitis. Dr. Arthur Krigsman is joining us as well today to
discuss his clinical findings of inflammatory bowel disorder in autistic children. He will share with us his initial findings as well
as discuss his research plans currently with his Institutional Review Board for approval.
Do the epidemiological and
case control studies, which the CDC has attempted to use to refute Dr. Wakefield’s
laboratory results, answer the autism-vaccine questions honestly? Epidemiologist Dr. Walter Spitzer is back today to
answer this question. What else is
needed to prove or disprove a connection?
Unfortunately, rather than
considering the preliminary clinical findings of Dr. Wakefield as a newly documented
adverse reaction to a vaccine, the CDC attempted to refute these clinical findings through
an epidemiological review. While
epidemiological research is very important, it cannot be used to disprove laboratory and
clinical findings. Valuable time was lost in
replicating this research and determining whether the hypothesis was accurate.
Officials at HHS have
aggressively denied any possible connection between vaccines and autism. They have waged an information campaign endorsing
one conclusion on an issue where the science is still out.
This has significantly undermined public confidence in the career public
service professionals who are charged with balancing the dual roles of assuring the safety
of vaccines and increasing immunization rates. Increasingly,
parents come to us with concerns that integrity and an honest public health response to a
crisis have been left by the wayside in lieu of protecting the public health agenda to
fully immunize children. Parents are
increasingly concerned that the Department may be inherently conflicted in its multiple
roles of promoting immunization, regulating manufacturers, looking for adverse events,
managing the vaccine injury compensation program, and developing new vaccines. Families share my concern that vaccine
manufacturers have too much influence as well.
How will HHS restore the public’s trust?
Access to the Vaccine Safety Datalink
(VSD)
One of the primary topics to
be discussed at this hearing is access to the Vaccine Safety Datalink. (VSD). To
help fill scientific gaps, the CDC formed partnerships with eight large health maintenance
organizations through an agreement with the American Association of Health Plans to
continually evaluate vaccine safety. This project is known as the Vaccine Safety Datalink
(VSD) and includes medical records on millions of children and adults. Up until this year, access to data from the VSD
has been limited to researchers affiliated with the CDC and a few of their handpicked
friends. This ‘good old boy’s
network” practice has predictably led to questions about the objectivity of the
research and the fairness of the results.
The VSD data should be made
available to all legitimate scientific researchers so that independent studies can be
conducted and results verified. This database
contains a wealth of data involving millions of patients over a ten-year period. If properly utilized, it can help researchers
study vitally important questions about the safety of vaccines, the effects of
mercury-based preservatives in childhood vaccines, and many other questions.
The Committee first raised this issue with the CDC two years ago. For two years the CDC delayed. Six months ago, we were informed that the CDC was
developing a plan to expand access to the database. Finally,
in February of this year, after a great deal of prompting from the Committee, Dr. Robert
Chen, Chief of Vaccine Safety and Development at the National Immunization Program,
informed Committee staff that the CDC had finalized its plan and that it was poised to put
it into effect. Under this plan, any
legitimate scientist could submit a proposal to the CDC to conduct research using VSD data
and access to the data would be provided along with some basic safeguards.
In preparation for today’s
hearing, Committee staff asked the CDC why the plan described to us in February had not
yet been put into effect. The staff was
informed that the plan had been put into effect. However,
there had been no public announcement. How
are researchers supposed to know about the availability of the data if there is no
announcement? It took two years of prodding
by this Committee to get the CDC to open up access to the database. For four months it appears that the CDC didn’t
inform anybody but this Committee of the data’s availability.
That doesn’t make it
appear that the CDC is making a good faith effort to open up this database. It looks to me like the CDC is trying to do the
bare minimum that they have to do to get us off their backs. That’s not acceptable. That’s why I insisted that Dr. Chen be here
today. I just want to ask him why they didn’t
tell anyone about the database being available. I’d
like to know how he expects researchers to use this data if nobody tells them it’s
available.
Dr. Roger Bernier is here
from the CDC to testify about these issues. He
is accompanied by both Dr. Chen, the creator of the VSD Project and Dr. Frank DeStefano,
the CDC official who is also a co-author of the MMR – IBD study. They are here to address our questions on the VSD
project and the vaccine-
autism research. The CDC employees are accompanied by Dr. Stephen
Foote of the National Institutes of Health and Dr. William Egan of the Food and Drug
Administration.
As representatives of the
people, we have a responsibility to ensure that our public health officials are adequately
and honestly addressing this epidemic and its possible links to vaccine injury.
I
look forward to hearing from our witnesses today. Our hearing record will remain open
until July 3.
I
now recognize the ranking minority member, Mr. Waxman for his opening statement.