Time Travel Research Center © 2005 Cetin BAL - GSM:+90 05366063183 - Turkey / Denizli
Radioactivity; fission and fusion; and nuclear energy
Introduction
This page provides a very brief introduction to radiation, radioactivity and nuclear energy. If you would like more information, then NIA has produced a set of four informative, educational pamphlets - the "Nuclear Power" series:
- Radiation and Nuclear Power
- Fission, Fusion and Safety and Nuclear Power
- Developing Energy and Nuclear Power
- Environment and Nuclear Power
So...What is radiation and radioactivity?
All matter is made up of atoms. All atoms are made up of three particles: protons, which have a positive electric charge; electrons, which are negatively charged; and neutrons, which have no electrical charge. The protons and neutrons combine to create the nucleus, which is the centre of an atom.
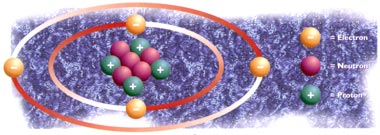
The number of protons defines which element it is; this is known as the element’s "atomic number". However, the elements atomic mass is equal to the number of protons plus the number of neutrons. All atoms of the same element have the same number of protons, but they do not have to have the same number of neutrons. In other words, atoms of the same element can have a different atomic mass. These different forms are called "isotopes". Most elements have several isotopes. For example, carbon-12 (C-12) is an isotope of carbon with an atomic mass of 12, whereas carbon-14 (C-14) is carbon with an atomic mass of 14. Isotopes have the same chemical properties, but often have slightly different physical properties, due to the differences in their mass.
Many naturally occurring isotopes are stable, which means that their nuclei remain the same and do not change spontaneously. However, in some cases, a particular isotope, or several isotopes even, of an element are unstable. Unstable isotopes want to become stable. They do this by spontaneously changing; the nucleus disintegrates, or "decays", altering either the number of protons or the number of protons and neutrons, until a stable form, but of a different element, is finally reached. In some cases, a particular unstable isotope will decay into another unstable isotope, which will itself then decay, and so on, until a stable isotope is finally reached. This chain of unstable elements is called a "decay chain". Decay chains can be very long or very short, depending on the particular isotope.
It takes energy to hold the protons and neutrons together in the nucleus. During the decay process, some of that energy, plus small particles (bits of the disintegrating nucleus), are given off or "radiated". This is why the word "radiation" has become used to describe the energy and small atomic particles released when unstable or "radioactive" isotopes ("radioisotopes") decay.
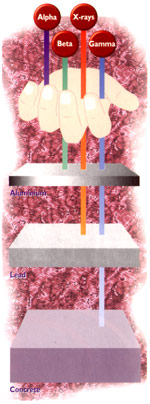 |
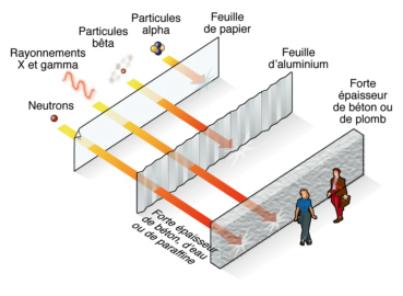
This type of radiation is actually more accurately described as "ionising" radiation, because these radiating particles and energy interact with the materials they strike to produce electrically charged particles, "ions", in a process known as "ionisation". There are several different forms of ionising radiation. The most important are alpha (a) particles , beta (b) particles, X-Rays and gamma (c)-rays. Alpha and beta radiation are tiny atomic particles travelling at high speeds. X- and gamma-rays are pure energy, like visible light, but far more energetic. Radiation can cause damage to living things, like the cells in our body, but this can also be a good thing. For example, radiotherapy treatment for cancer works by using radiation to destroy the cancer cells. Radiation occurs naturally. We are surrounded by radiation. It comes from all materials on Earth, including rocks, soil, air and water, as well as from the sun and outer space "cosmic radiation"). Radioactivity was discovered by Henri Becquerel in 1896, the year after Wilhelm Rontgen discovered X-rays. There are now nine known naturally occurring radioactive elements. Some of the best known naturally occurring radioactive elements are uranium, carbon-14 (that is, an isotope of carbon with a mass of 14; the most common, stable, isotope of carbon is carbon-12), radium (once used to make luminous dials for clocks and watches) and radon. |
There are also a large number of man-made radioactive elements and radioactive isotopes of non-radioactive natuarally occurring elements, the most well known two being plutonium and tritium (or hydrogen-3), a radioactive form of hydrogen.
Fission and Fusion: What is nuclear energy?
The term "nuclear" or "atomic" energy has arisen because it makes use of the energy stored within the atom or, more accurately, within the nucleus of an atom. There are two ways of releasing and harnessing this nuclear energy - "nuclear fission" and "nuclear fusion".
Nuclear fission
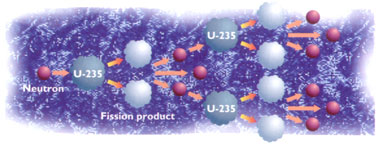
Nuclear fission is the splitting of the nucleus of a radioactive element into two smaller fragments. The nucleus is caused to fission by striking it with a neutron. The fissioning of the nucleus releases energy; this is referred to as "nuclear energy". If the radioisotope used is uranium-235, then the fission reaction, as well as releasing energy, also releases three more neutrons. Therefore, if there are other uranium-235 nuclei around, then one of them might, in turn, be struck by one of these three new neutrons, causing it to fission in turn, generating yet more neutrons.......and so on. In other words, a chain reaction can be generated, which releases a lot of energy.
Nuclear fission power is based on the safe harnessing of this energy. In simple terms, the nuclear energy (basically heat) released is used either directly or indirectly to generate steam, which is then used to drive steam turbines, which generate electricity.
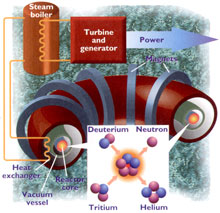 |
|
Nuclear fusion Nuclear fusion is the opposite of fission. Fusion involves forcing nuclei to fuse, or join, together, rather than splitting them apart. Stars, such as the sun, are powered by fusion reactions. Unfortunately, because nuclei are positively charged, they naturally want to repel each other (like magnets of the same pole). Therefore, to force nuclei together initially requires a lot of energy. Fusion reactions only occur at very hightemperatures (millions of degrees centigrade) and pressures. |
Scientists have been able to achieve fusion reactions, using devices such as the Joint European Torus (JET), but it is not yet practical to use nuclear fusion to generate electricity. However, if it could be made practical, then nuclear fusion offers a lot of benefits. Therefore, a lot of research is still being carried and it is hoped that, one day, nuclear fusion will become an alternative source of energy production.
© Copyright 2003 - Nuclear Industry Association
Alıntı: http://www.niauk.org/article_10.shtml
Hiçbir yazı/ resim izinsiz olarak kullanılamaz!! Telif hakları uyarınca bu bir suçtur..! Tüm hakları Çetin BAL' a aittir. Kaynak gösterilmek şartıyla siteden alıntı yapılabilir.
The Time Machine Project © 2005 Cetin BAL - GSM:+90 05366063183 -Turkiye/Denizli
Ana Sayfa /İndex /Roket bilimi / ![]() E-Mail /CetinBAL /Quantum Teleportation-2
E-Mail /CetinBAL /Quantum Teleportation-2
Time Travel Technology /Ziyaretçi Defteri / Duyuru / UFO Technology