Nuclear Fission
 |
The Enrichment Process
The enrichment process is
complicated due to the fact that the uranium isotopes are practically
chemically identical. This means that the different isotopes cannot be
separated with just an easy chemical reaction. The isotopes must be
separated by exploiting the little mass difference between the isotopes.
There are many different methods used to separate these isotopes a
couple of these are the commercial-gaseous diffusion process and the
centrifuge process. Another method of separation is the Electro
Magnetic Separation process.
This enrichment method was created by
E.O. Lawrence. It involves passing uranium ions through a magnetic
field which causes the U235
ions to separate and take a different path than the U238 ions.
Following this collectors are used at the other end of the semi-circle
to capture the separated U235.
Fission of U235

There were two types of atom bombs created one was called the "Little Boy" and the other was titled the "Fat Man."
The Little Boy
The "little boy" atom bomb was designed to be a gun type bomb. They call it a gun type bomb due to the fact that the bomb shoots a mass of uranium 235 into another mass of uranium 235 to create a supercritical mass.

www
Once the supercritical mass is formed the initiator emits a burst of neutrons creating the fission chain reactions. These chain reactions continue until the energy released becomes so big that the bomb blows itself up.
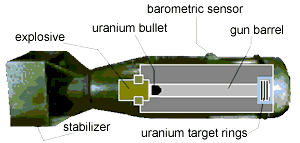
The Fat Man:
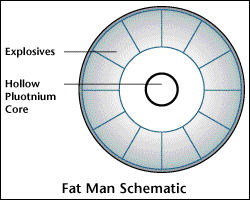
The Fat Man atom bomb was created due to the fact that plutonium 239 has such a fast spontaneous fission rate. A new design was needed to facilitate such an isotope. The Little Boy is too slow to work with such an isotope. So, Seth Neddermeyer created an idea to use explosive charges to compress a small sphere of plutonium quickly to a required density that will make the plutonium go critical and produce a nuclear explosion. Thus, the Fat Man atom bomb was born.
Building the Bomb
Fission: The Atomic Bomb
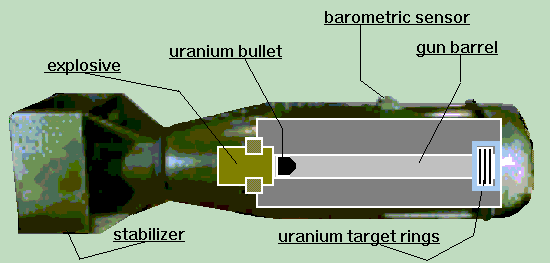
The atomic bombs that were used on Hiroshima and Nagasaki derived their explosive power via a fission chain reaction. The fuel used for Little Boy, the bomb used on Hiroshima, was enriched uranium. Fat Man, used on Nagasaki, was fueled by plutonium.
Below is a sketch of the kind of uranium bomb used on Hiroshima. The explosive drives the uranium "bullet" into the secondary mass of uranium to create a SuperCritical mass.
Fusion: The Nuclear Bomb
"In thermonuclear weapons, radiation from a fission explosive can be contained and used to transfer energy to compress and ignite a physically separate component containing thermonuclear fuel."
The 3 basic concepts of thermonuclear devices, U.S. DOE, Sept 1980, Duane Sewell, Assistant Secretary of Energy for Defense, Official Declassification Act.
Fusion Fuel
The forms of Hydrogen used in a fusion reaction -- H2 (deuterium) and H3 (tritium) -- are gases at room temperature, so their storage in a weapon would be difficult. Instead, a substance called lithium deuteride (Li6D or Li7D) is used.
Lithium
The lightest of metals, lithium is only half as dense as water. Lithium is found combined with other elements in igneous rocks and mineral spring water. Lithium has several isotopes, ranging from Li5 to Li9. Li6 and Li7 are used in weapons, and are naturally occuring. Li5, Li8, and Li9 are man-made radioisotopes.
Li6 is present as 7.5% of all naturally occuring Li. Separation methods include electrolysis, distillation, chemical exchange, or EM methods. Li bonds with H to form the solid Li6D. This material is a whitish, slightly-blue powdery light salt-solid (which is extremely hygroscopic) at room temperature. It is made by heating metal lithium in a vessel, into which deuterium gas is injected. It is then pressed and shaped into a ceramic.
Li6D in the Fusion Reaction
When a neutron is absorbed by a molecule of litium deuteride (Li6D), the molecule breaks up into a He, H2 (deuterium) and H3 (tritium). The deuterium can then react with the tritium in fusion. This releases enormous amounts of energy, much greater than you would get in a fission reaction. The end products include a free neutron and a helium atom. Schematically:
Li6 + n -> He4 + T + 4.7 MeV
then
D + T -> He4 + n + 17.6 MeV.
n + U-238 -> neutrons + fission products + energy
These reactions occur in under 1/10-6 secs.
Li6D in the Bomb
In order to begin a fusion reaction in a thermonuclear weapon, the lithium deuteride solid must be compressed to 15-30 times its original uncompressed density at RTP (15lbs/foot^3).
Compression is achieved by a smaller fission reaction, which forces the Li6D fuel together.
[image: showing fission reaction forcing fusion fuel together]
Compression is needed to:
By packing the molecules closer together, you increase the probability of fusion occuring. Compressing the molecules paves the way to overcoming the electrostatic repulsion of the H atoms in the Li6D.
A faster rate of fusion produces a larger chain reaction. The reaction rate is proportional to the square of the fuel density -- in other words, the more compressed the fuel is, the shorter the reaction time will be. Increase the density by a factor of 30, for example, and the rate of fusion increases by a factor of 900.
Hiçbir yazı/ resim izinsiz olarak kullanılamaz!! Telif hakları uyarınca bu bir suçtur..! Tüm hakları Çetin BAL' a aittir. Kaynak gösterilmek şartıyla siteden alıntı yapılabilir.
The Time Machine Project © 2005 Cetin BAL - GSM:+90 05366063183 -Turkiye/Denizli
Ana Sayfa /İndex /Roket bilimi / ![]() E-Mail /CetinBAL /Quantum Teleportation-2
E-Mail /CetinBAL /Quantum Teleportation-2
Time Travel Technology /Ziyaretçi Defteri / Duyuru / UFO Technology