Time Travel Research Center © 2005 Cetin BAL - GSM:+90 05366063183 - Turkey/Denizli
The Atomic
Nucleus
Isotopes
 |
Same number of protons, but different numbers of neutrons. Electrical and |
Quarks and the Strong Force
 |
Quarks are responsible for the nuclear force between nucleons. The nuclear force is also known |
Unstable Nuclei
 |
Neutrons 1-4 provide strong forces of attraction for the indicated proton, but other neutrons are too far away to help balance the longer- range forces of repulsion provided by the many protons to the right of the proton. The larger the nucleus, the more unstable it is. |
Uranium Decays via Alpha-Particle Emission
 The first particle ejected from an unstable nucleus was called an alpha particle because alpha is the first letter of the Greek alphabet. It's now known to consist of two protons and two neutrons, which is the same as a helium nucleus. |
Balancing Nuclear Disintegration Equations
 Total number of nucleons is conserved: 238 =====> 234 + 4 Total charge is conserved: 92 =====> 90 + 2 |
Reaching Stability Through Gamma Ray Emission
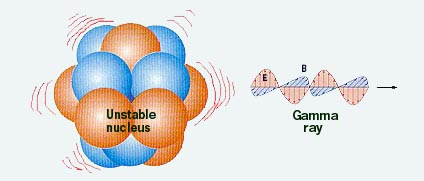 |
Nuclei with excess energy emit gamma-rays, which are extremely short- wavelength electro- magnetic waves, i.e., very high energy photons. |
Blocking Radiation
 |
Alpha particles are 8,000 times as heavy as beta particles. Paper or clothing will block alpha particles, while beta particles require a few sheets of aluminum foil. Gamma radiation is extremely dangerous--a thousand times more potent than xrays. |
Radioactivity in Radium Killed Marie Curie
 88Ra226 ======> 86Rn222 + 2He4 |
 |
Marie Curie
 Marie Sklodowska Curie (1867-1934) |
 Lithograph entitled "Radium" appeared in the December 22, 1904 issue of Vanity Fair. |
Marie and Pierre Curie isolated 1/30 ounce of radium from one ton of uranium ore. Marie died from radiation-induced leukemia. The pages of her lab notebook were later found to be contaminated with radioactive fingerprints. |
Uranium Dating
 |
 |
Transmuting Elements
 |
Transmuting Uranium to Neptunium
 Neutron enters nucleus and is transformed into a proton and an electron (which leaves the nucleus). |
Albert Einstein and Mass-Energy Equivalence
 Albert Einstein (1879-1955) |
 Mass is energy: E = mc2 Energy is mass: m = E /c2 |
When a uranium nucleus splits, the mass of the remnants is less than the original mass. The difference appears as light, heat, and kinetic energy. |
Nuclear Fission
 |
Average number of neutrons released is 2.5. Combined kinetic energy of C + O2 => CO2 |
(Slow neutrons cause fission)
 |
Slow neutrons are required. A chain reaction occurs if Neutrons can be slowed by One pound of U-235, if |
Cadmium Control Rods Absorb Neutrons
 Enrico Fermi supervised construction of the world's first nuclear reactor. |
 Cadmium is a good absorber of neutrons. |
World's First Controlled Nuclear Chain Reaction
 Handball court under the bleachers at the University of Chicago, 1942. Uranium-235 is at the center of the stack of graphite blocks; the carbon acts as a moderator, slowing neutrons. |
The Manhattan Project
 Oak Ridge, Tennessee. 60,000 workers worked for three years to separate 2 kilograms of uranium-235 from uranium-238. |
Critical Mass
 The smaller the sphere, the greater the ratio of surface area to volume, and the greater the percentage of neutrons which escape the sphere before causing fission. Critical mass--or, critical size--is that mass value at which an average of more than one neutron per fission is used to cause another fission. |
Fission Bomb Idealized
 |
Two sub-critical masses are smashed together to create a super-critical mass. |
Trinity Test Site
 5:30 am, July 16, 1945, Alamogordo, New Mexico. |
|
The First Atomic Bomb
 "Little Boy", two feet in diameter, ten feet long, 9000 pounds, dropped on Hiroshima, Japan, was a uranium bomb, equivalent to 20,000 tons of explosive. |
Little Boy and Fat Man
 Little boy is on the left. |
The Reactor Vessel
 |
The water in the reactor vessel has three purposes. The water, being composed of Water also acts to remove heat |
A Nuclear Reactor
 |
Heat generated by fission in uranium rods creates steam which turns turbine blades connected to a coil of wire in magnetic field. |
Discoverer of Nucleus Was Wrong about Nuclear Power
 |
 Rutherford at 1911 Solvay Conference |
Fusion
 Fusion is the opposite of fission. Deuterium must be moving extremely fast to fuse. |
Fusion in Stars
 10 million degrees at the core causes fusion of hydrogen into helium. |
Deuterium-Tritium Fusion
 |
Laser evaporates D-T, creating a "plasma" of charged particles which push away from one another. The reaction force compresses and heats core. |
Thermonuclear Weapons
| The hydrogen bomb uses an atomic bomb as the heat
source to to fuse hydrogen into helium. The so-called H-bomb is vastly more destructive than fission bombs. The Hiroshima bomb had had explosive power of about 20,000 tons of TNT; H-bombs commonly have 50-500 times the power (1-10 megatons). Several hydrogen warheads are mounted on some of our missiles, and each warhead, upon re-entering the atmosphere, is independently targeted at a different city. |
Mass is Energy and Energy is Mass
 |
The work done per nucleon adds energy to each nucleon, which appears as increased mass. Nucleons broken out of the nucleus weigh more outside than they do inside. |
Fusion versus Fission
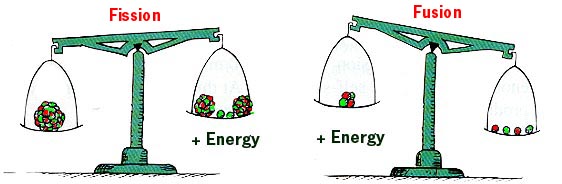 If the final products have less mass than the reactants, energy is released. |
Fission versus Fusion
 |
Hiçbir yazı/ resim izinsiz olarak kullanılamaz!! Telif hakları uyarınca bu bir suçtur..! Tüm hakları Çetin BAL' a aittir. Kaynak gösterilmek şartıyla siteden alıntı yapılabilir.
The Time Machine Project © 2005 Cetin BAL - GSM:+90 05366063183 -Turkiye/Denizli
Ana Sayfa /
index /Roket bilimi /![]() E-Mail /CetinBAL/Quantum Teleportation-2
E-Mail /CetinBAL/Quantum Teleportation-2
Time Travel Technology /Ziyaretçi Defteri /UFO Technology/Duyuru
